
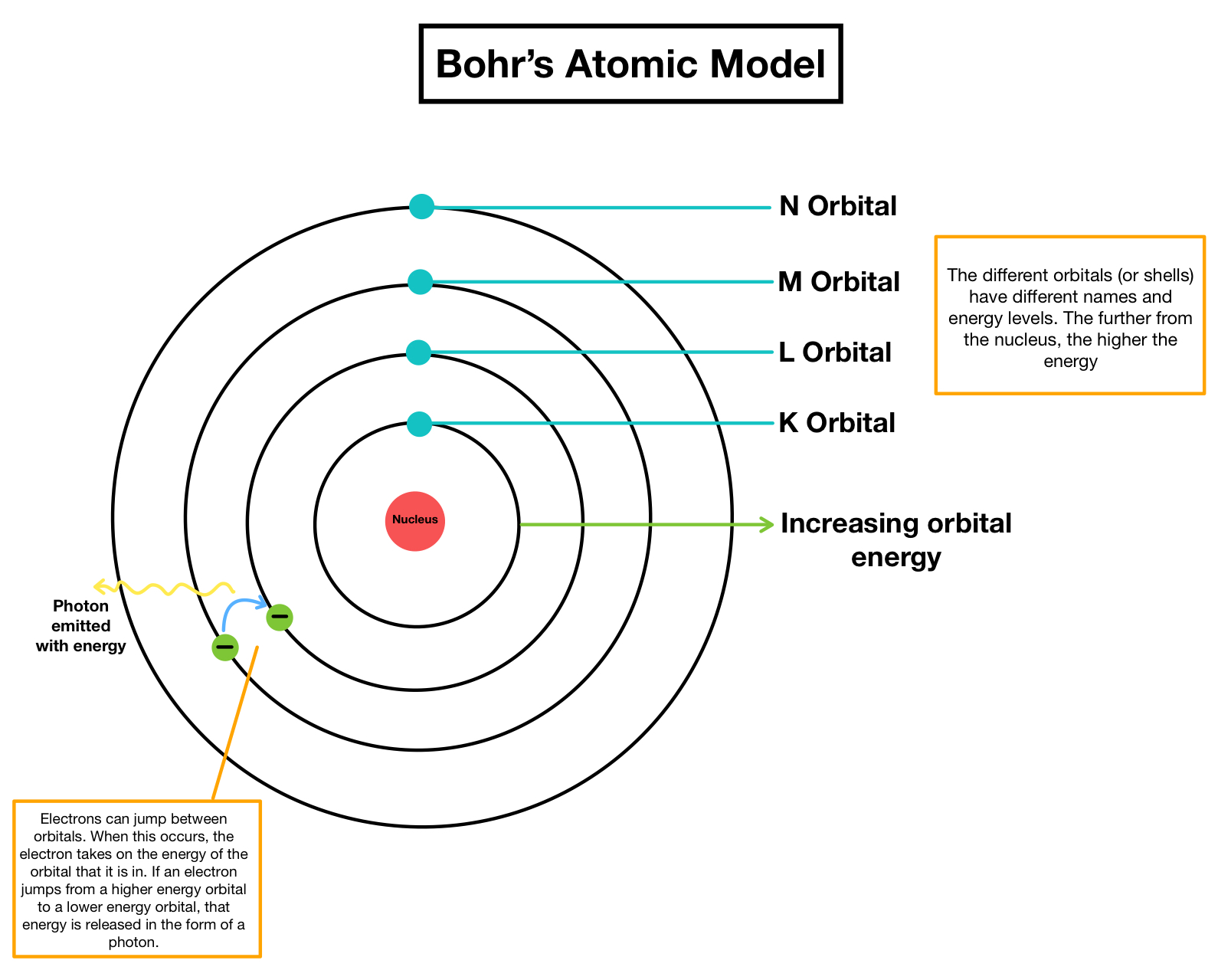
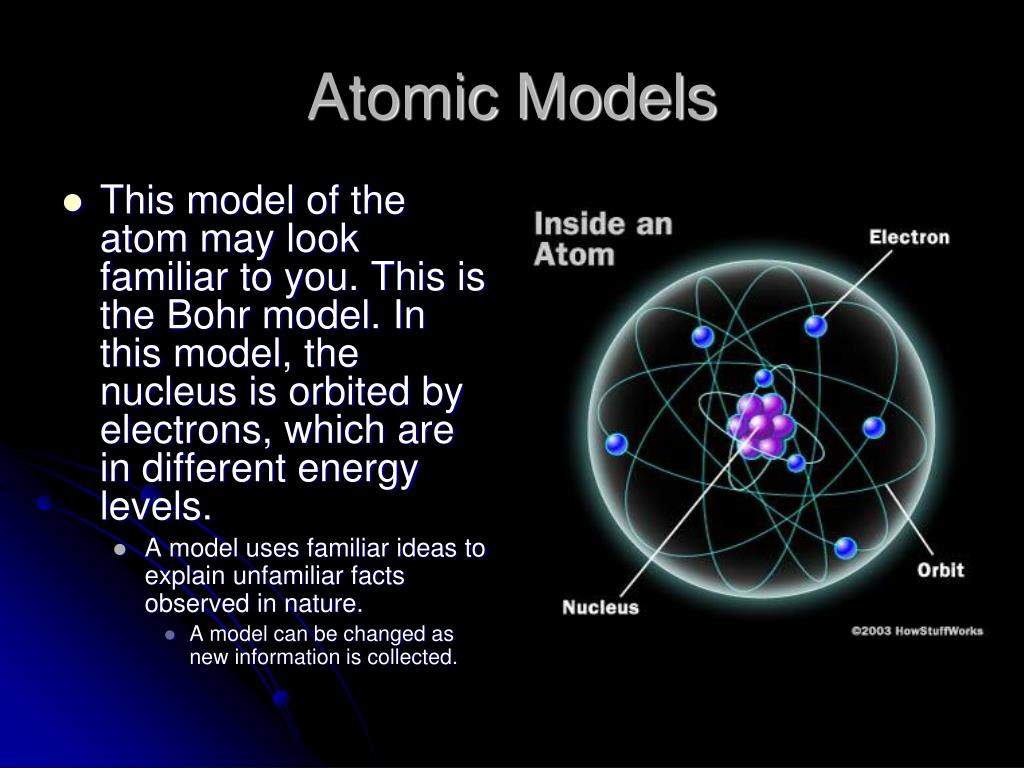
However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner. Within these regions, there are higher and lower probabilities of finding an occupying. An electron in an orbit is a self-accelerating electrically charged particle, and according to the laws of physics such particles must emit electromagnetic. Therefore, the modern atomic theory and model present the electron in cloud-like orbitals around the nucleus. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. Rutherford’s atom model was another big step forward in the development of atomic theory, however there were inherent problems with it as it violated fundamental principles of physics. For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Thus, the Bohr model for heavier atoms described electron shells. It wasnt until the 1980s that chemists could see individual atoms. For a couple thousand years, humans could only speculate on the structure and other properties of the smallest unit of matter. Once the level was full, additional electrons would be bumped up to the next level. 1: Since ancient Greek times, philosophers and scientists have tried to figure out what an atom looks like. 3.1: Atomic Theory is shared under a not declared license and was authored, remixed. Each element has its own atomic number, which is equal to the number of protons in its nucleus. Atoms themselves are composed of protons, neutrons, and electrons. Bohr believed each electron orbit could only hold a set number of electrons. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. More electrons were required to cancel out the positive charge of all of these protons. Isotopes of an element contain different numbers of neutrons. The quantum mechanical model of the atom comes from the solution to. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms.
The electrons are embedded in this sphere so as to give the most stable electrostatic arrangement. In 1926, Austrian physicist Erwin Schrödinger (1887-1961) used the wave-particle duality of the electron to develop and solve a complex mathematical equation that accurately described the behavior of the electron in a hydrogen atom. It was based on knowledge of the recently discovered negatively charged subatomic particles called electrons. He proposed that an atom is shaped like a sphere with a radius of approximately 10-10 m, where the positive charge is uniformly distributed. Thompson posited the plum pudding, or raisin bun, model of atomism. Heavier atoms contain more protons in the nucleus than the hydrogen atom. Thomson proposed the first of many atomic models to come.
